Ultime Notizie
Bomi Group secures sustainable certification in Brazil and Italy
We are proud to announce for the first time the…
Bomi Group, “a UPS company”, inaugura uno delle più grandi facility del settore healthcare a livello europeo
Bomi Group, "a UPS company" con headquarter a Spino D’Adda…
UPS completa l’acquisizione di Bomi Group, multinazionale specializzata nella logistica healthcare
L'acquisizione accresce il portfolio di servizi tra cui quelli della…
In primo piano
"Not only logistic!"
ma partner dei nostri clienti
La Missione di Bomi Group è quella di disegnare soluzioni logistiche a supporto della supply chain del settore Healthcare, superando il concetto di logistica standard.

BOMI GROUP HEADQUARTERS
Inaugurata l’estensione dell’Headquarter Bomi Group in Italia. Nuovi spazi, automazione e sicurezza le parole chiave del progetto
BOMI GROUP HEADQUARTERS

Sustainable Logistics
BOMI Group ottiene il marchio "Sustainable Logistics" e porta la sostenibilità nella filiera della salute
Sustainable Logistics

Bomi Health Carrier
Soluzioni offerte
Servizio informativi e tracking delle consegne
Home Care Service
White Gloves service
Bomi Health Carrier

Officina Farmaceutica
Applicazione Bollini Ottici
Sostituzione foglietti illustrativi e astucci
Stampa dei dati variabili
Riconfezionamento secondario
Officina Farmaceutica
Il nostro Network Globale
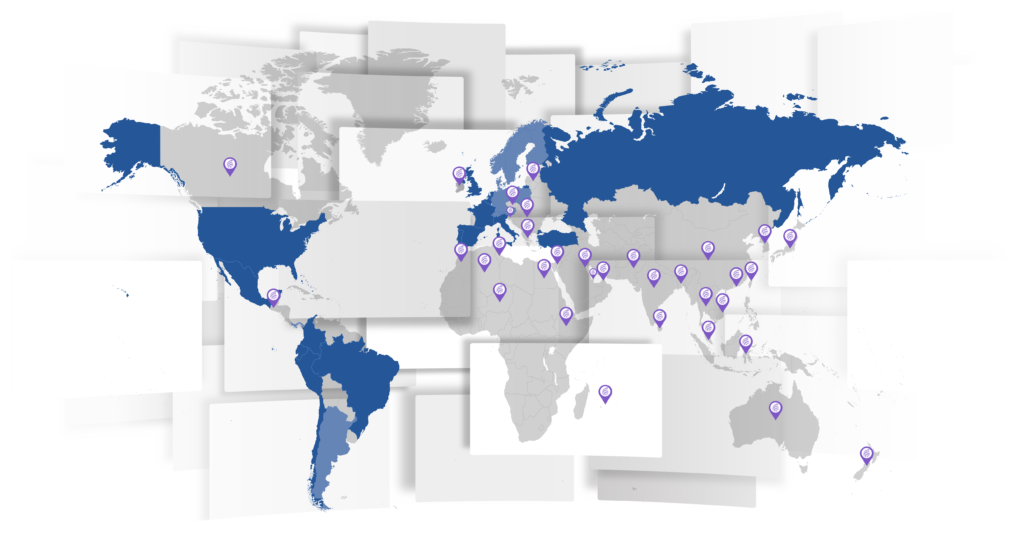
going much beyond the pure logistic concept















